KalVista Confirms Mechanism of Action of Potential HAE Therapy KVD900
Written by |

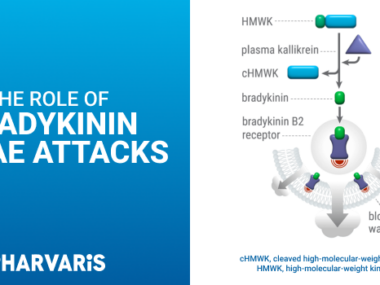
KVD900, a potential new approach for treating hereditary angioedema, prevents the breakdown of kininogen and subsequent buildup of bradykinin — the underlying cause of the disease, KalVista Pharmaceuticals announced.
The company confirmed this mechanism of action using a new test that measures the amounts of high molecular weight kininogen in the plasma.
The results were recently shared in the poster titled, “KVD900 protects high molecular weight kininogen from ex vivo plasma kallikrein-mediated cleavage in plasma from patients with hereditary angioedema (HAE): Results from capillary-based immunoassay.”
The poster was presented at the European Academy of Allergy and Clinical Immunology (EAACI) Congress, held May 26-30 in Munich, Germany.
Hereditary angioedema (HAE) is a potentially life-threatening disease that in most cases is caused by mutations in the C1-inhibitor gene. The defects lead to excess bradykinin, a peptide that causes blood vessels to dilate and is responsible for most acute angioedema symptoms.
Bradykinin is generated from another molecule, called kininogen. Therefore, changes in the blood levels of kininogen have been proven useful, not only as a biomarker for HAE attacks but also to assess the effectiveness of therapies.
To date, no automated ways existed for measuring kininogen rapidly and reliably. KalVista developed a semi-automatic, high-throughput immunoassay that measures this protein in the blood plasma.
The immunoassay, which uses antibodies against kininogen placed inside tiny capillaries, was able to detect kininogen in blood samples five times faster and with simpler procedures than traditional methods.
The company’s leading therapy, KVD900, was developed to inhibit plasma kallikrein, the protein that helps kininogen cleave onto bradykinin. Using the new test in six angioedema patients, investigators confirmed the medicine’s mechanism of action.
KVD900 prevented the breakdown of kininogen in a dose-dependent manner in both HAE patients and in healthy volunteers.
“We are pleased to have developed a proprietary immunoassay to characterize the effects of our novel orally-available plasma kallikrein inhibitor, KVD900, in development for treatment of hereditary angioedema, or HAE,” Andrew Crockett, the CEO of KalVista, said in a press release.
“Using this method, we are able to demonstrate that KVD900 protects high molecular weight kininogen from plasma kallikrein mediated cleavage in HAE and control plasma. We look forward to providing an update on our HAE portfolio in the next few months,” he added.